Dear All,
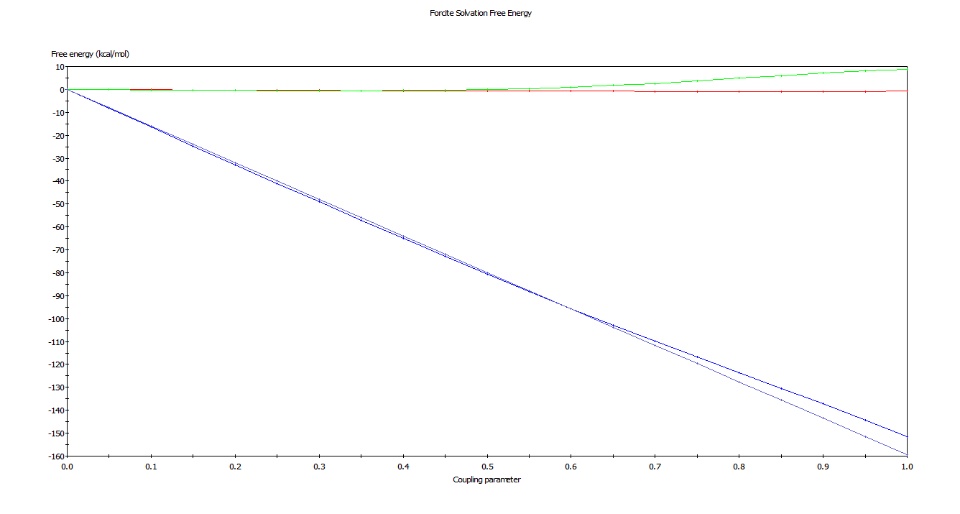
I want to calculate the solvation free energy of a cation in a solid-aqueous-solid sandwich-like system. I've already finished the MD equilibration. I calculated the solvation free energy, I set the euqilibration and production steps both as 1000000, the time steps as 1fs. It can finish very quickly, in minutes, even though my server is not super computer. However, I checked the energy chart, the energy wasn't stable at the end of calculation. I attached the chart. The dark blue line is the total energy.
I also tried to calcualte the solvation energy of the cation which was inserted into an amorphous cell with water molecules and the cation was charge balanced with another ion. But the task failed after several days of calculation, it couldn't enter the production phase.
Any suggestion would be appreciated!
Regards,
Jingjing
I want to calculate the solvation free energy of a cation in a solid-aqueous-solid sandwich-like system. I've already finished the MD equilibration. I calculated the solvation free energy, I set the euqilibration and production steps both as 1000000, the time steps as 1fs. It can finish very quickly, in minutes, even though my server is not super computer. However, I checked the energy chart, the energy wasn't stable at the end of calculation. I attached the chart. The dark blue line is the total energy.
I also tried to calcualte the solvation energy of the cation which was inserted into an amorphous cell with water molecules and the cation was charge balanced with another ion. But the task failed after several days of calculation, it couldn't enter the production phase.
Any suggestion would be appreciated!
Regards,
Jingjing

